Abstract:This study evaluates the photocatalytic performance of Ag–ZnO nanoparticles for the degradation of Rhodamine B (RhB), a widely encountered organic dye pollutant in wastewater. The synthesized Ag–ZnO photocatalyst was characterized using SEM/EDS to elucidate its surface morphology and elemental composition. Experimental results demonstrate a high degradation efficiency, with optimal conditions identified at a catalyst dosage of 0.06 g in a neutral medium (pH 7). Under these conditions, 78% of RhB at an initial concentration of 50 mg/L was degraded after 120 minutes of UV irradiation. Kinetic analysis indicates that the degradation process follows a pseudo-first-order model, with a strong correlation coefficient (R² = 0.9901), confirming excellent agreement between experimental data and the proposed kinetic model. The apparent reaction rate constant was calculated to be 0.0123 min⁻¹, reflecting the relatively strong photocatalytic activity of the synthesized material compared with previously reported systems. These findings underscore the potential of Ag–ZnO nanoparticles as an effective and promising photocatalyst for the treatment of dye-contaminated wastewater.Keywords: Ag-ZnO, photocatalytic degradation, Rhodamine B, UV light, kinetic model, wastewater treatment.
1. Introduction
The textile dyeing industry is one of the major contributors to industrial wastewater pollution due to the discharge of large volumes of colored effluents containing recalcitrant organic compounds. Among these pollutants, Rhodamine B (RhB) is widely recognized as a hazardous xanthene dye because of its high chemical stability, toxicity, and poor biodegradability. Conventional treatment methods, such as coagulation, adsorption, membrane filtration, and biological treatment, often suffer from limited removal efficiency, high operating costs, or the generation of secondary waste (Forgacs et al., 2004; Robinson et al., 2001).
Advanced oxidation processes (AOPs), particularly heterogeneous photocatalysis, have attracted considerable attention as environmentally friendly alternatives for dye removal. Semiconductor photocatalysts such as TiO₂ and ZnO can generate electron–hole pairs under light irradiation, producing reactive oxygen species capable of oxidizing organic contaminants into less harmful compounds (Fujishima & Honda, 1972; Herrmann, 1999). ZnO is especially attractive due to its low cost, high photosensitivity, and strong oxidation potential; however, its practical application is limited by rapid electron–hole recombination and its primary activity in the UV region (Herrmann, 1999).
To overcome these limitations, surface modification of ZnO with noble metals has been widely explored. Silver (Ag) can act as an electron trap, suppressing charge recombination and enhancing photocatalytic efficiency, while also facilitating interfacial charge transfer. Consequently, Ag/ZnO nanocomposites have been proposed as promising photocatalysts for improving the degradation of dye pollutants (Zhai et al., 2015). This study aimed to synthesize an Ag/ZnO nanocomposite and evaluate its photocatalytic performance in degrading RhB under UV irradiation. The influence of pH and catalyst dosage was examined, and the reaction kinetics were analyzed.
2. Materials and methods
2.1. Chemicals
The chemicals used in this study included zinc sulfate heptahydrate (ZnSO4·7H2O), cetyltrimethylammonium bromide (CTAB), silver nitrate (AgNO3), hydrazine hydrate (N2H4·H2O, 80%), ammonium hydroxide (NH4OH), sodium chloride (NaCl), distilled water, and Rhodamine B (RhB). All reagents were used as received without further purification.
2.2. Synthesis of ZnO and Ag/ZnO
ZnO nanoparticles were synthesized via a precipitation-assisted method. Briefly, 3.18 g of ZnSO4·7H2O and 4.02 g of CTAB were added into a 500 mL beaker at a molar ratio of 1:1. Then, 100 mL of distilled water was added, and the mixture was stirred at 700 rpm while heated at 60 C until a clear and homogeneous solution was obtained. Subsequently, an additional 300-400 mL of distilled water was added, and the suspension was maintained in a water bath at 80 C for 5 h to promote nucleation and crystal growth. After cooling to room temperature, NH4OH was added to adjust the pH to approximately 10, inducing precipitation of zinc hydroxide, which then converted into ZnO upon further processing. The precipitate was washed repeatedly with distilled water to remove residual CTAB and impurities, filtered, and dried at 80 C for 6-8 h to obtain fine ZnO powder.
Ag/ZnO was prepared by chemical reduction of Ag+ adsorbed on ZnO. First, 0.3 g of ZnO was dispersed in 20 mL of distilled water by magnetic stirring at 600 rpm and ultrasonication for 15 min. Then, 70 mL of AgNO3 solution was added, and the mixture was stirred at 700 rpm for 2 h to allow adsorption of Ag+ onto the ZnO surface. The solid was filtered and washed twice with distilled water to remove excess AgNO3. Next, the material was re-dispersed in 30 mL of distilled water, and 0.2 mL of hydrazine hydrate was slowly added during stirring to reduce Ag+ to metallic Ag nanoparticles on the ZnO surface. The resulting product was washed several times, filtered, and dried at 70 C for 6 h, yielding a light gray Ag/ZnO powder.
2.3 Photocatalytic Degradation Experiments
Batch photocatalytic experiments were carried out in a 250 mL beaker placed on a magnetic stirrer and irradiated with a UV lamp. A 100 mL RhB solution (50 mg/L) was used for each experiment. The effects of pH (4, 5, 6, and 7) and catalyst dosage (0.04, 0.06, 0.08, and 0.10 g) were investigated. Samples were collected from 0 to 120 min and analyzed by UV-Vis spectroscopy at 552 nm to determine the residual RhB concentration.
![]()
where: Ce (mg/L) is the concentration at equilibrium; C0 (mg/L) is the initial concentration.
3. Results and discussion
3.1. Morphological Characteristics
SEM images (Fig.1) showed that pristine ZnO consisted mainly of short rod-like and prism-like nanostructures with relatively uniform particle distribution. This morphology is advantageous for maintaining photocatalytic activity due to effective charge separation and transfer. In contrast, Ag/ZnO exhibited rough, platelet- or flower-like nanostructures with wrinkled surfaces and particle sizes mainly below 200 nm. The rougher texture and more irregular surface of Ag/ZnO likely increased the available active sites for photocatalytic reactions. Although some aggregation was observed, the material still maintained a large effective contact area. These morphological features suggest that Ag deposition modified the ZnO surface in a way that is beneficial for photocatalytic degradation.
Figure 1. SEM image of ZnO and Ag/ZnO.
![]()
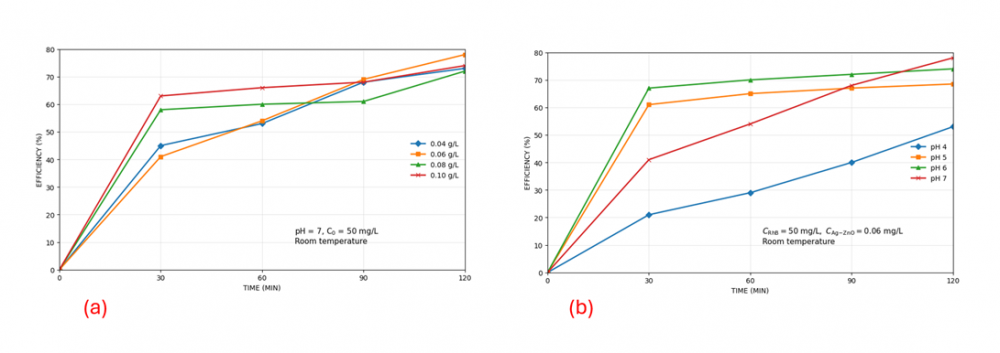
3.3. Effect of pH
Fig 2 (a) Solution pH significantly influenced the photocatalytic degradation efficiency of RhB. Under acidic conditions (pH 4-5), the degradation efficiency was lower, reaching only around 53.36% at pH 4 after 120 min. In contrast, higher efficiencies were obtained at near-neutral pH, with 74.79% at pH 6 and 78.10% at pH 7 after 120 min. The improved performance near neutral pH can be attributed to more favorable surface charge interactions between the catalyst and RhB, as well as enhanced generation of reactive oxygen species such as hydroxyl radicals and superoxide radicals. Therefore, pH 7 was identified as the optimum pH for RhB degradation in this system.
3.4. Effect of catalyst dosage
Fig2 (b). Catalyst dosage also played a crucial role in the photocatalytic process. Increasing the amount of Ag/ZnO from 0.04 g to 0.06 g improved the degradation efficiency from 72.92% to 78.10% after 120 min. However, further increasing the dosage to 0.08 g and 0.10 g did not improve the process and instead led to lower final efficiencies of 71.49% and 73.53%, respectively. This behavior suggests that excessive catalyst loading may cause particle aggregation and light scattering or shielding effects, reducing light penetration and limiting the activation of photocatalytic sites. Accordingly, 0.06 g was considered the optimal catalyst dosage under the conditions studied.
Figure 2. (a) Effect of pH; (b) Effect of Catalyst Dosage
3.5. Photocatalytic kinetics
The kinetic data fitted well to the pseudo-first-order Langmuir-Hinshelwood model, with the following equation: ln(C0/C) = 0.0123t + 0.0454. The apparent rate constant was k = 0.0123 min-1, and the correlation coefficient was R2 = 0.9901, indicating a strong agreement between the experimental data and the proposed kinetic model. The calculated half-life (t1/2) was approximately 56.35 min, while the estimated times required to reach 90% and 99% degradation were 187.2 min and 374.4 min, respectively. The non-zero intercept suggests that initial adsorption or other surface phenomena may have contributed to the degradation profile. Nonetheless, the obtained rate constant indicates that the synthesized Ag/ZnO exhibited reasonably good photocatalytic activity compared with previously reported studies.
4. Conclusions
Ag/ZnO nanocomposite was successfully synthesized through a precipitation-assisted preparation of ZnO followed by adsorption-reduction deposition of Ag onto the ZnO surface. SEM analysis confirmed that Ag modification altered the morphology of the material, producing rougher and more irregular nanostructures that are favorable for photocatalysis. Under UV irradiation, the synthesized Ag/ZnO effectively degraded Rhodamine B in aqueous solution. The optimum conditions identified in this study were pH 7 and a catalyst dosage of 0.06 g in a 100 mL reaction system, under which the degradation efficiency reached 78.1% after 120 min. Kinetic analysis showed that the photocatalytic degradation followed an apparent pseudo-first-order Langmuir-Hinshelwood model, with a rate constant of 0.0123 min-1 and R2 = 0.9901. These results indicate that Ag/ZnO is a promising photocatalyst for the treatment of dye-containing wastewater and may serve as a potential candidate for further development in environmental remediation applications.
Acknowledgment:
The author would like to greatly acknowledge the support of time and facilities from the Ho Chi Minh City University of Technology, Vietnam National University - Ho Chi Minh City for this study.
References:
Forgacs E., Cserháti T., & Oros G. (2004). Removal of synthetic dyes from wastewaters: A review. Environment International, 30(7), 953–971. https://doi.org/10.1016/j.envint.2004.02.001
Fujishima A., & Honda K. (1972). Electrochemical photolysis of water at a semiconductor electrode. Nature, 238(5358), 37–38. https://doi.org/10.1038/238037a0.
Herrmann J. M. (1999). Heterogeneous photocatalysis: Fundamentals and applications to the removal of various types of aqueous pollutants. Catalysis Today, 53(1), 115–129. https://doi.org/10.1016/S0920-5861(99)00107-8.
Robinson T., McMullan G., Marchant R., & Nigam P. (2001). Remediation of dyes in textile effluent: A critical review on current treatment technologies with a proposed alternative. Bioresource Technology, 77(3), 247–255. https://doi.org/10.1016/S0960-8524(00)00080-8.
Zhai H. J., Wang L. J., Sun D. W., Han D. L., Qi B., Li X. Y., Chang L. M., & Yang J. H. (2015). Direct sunlight responsive Ag–ZnO heterostructure photocatalyst: Enhanced degradation of rhodamine B. Journal of Physics and Chemistry of Solids, 78, 35–40. https://doi.org/10.1016/j.jpcs.2014.11.004.
Tổng hợp vật liệu xúc tác quang Ag/ZnO ứng dụng phân hủy Rhodamine B
Đặng Viết Hùng
Khoa Môi trường và Tài nguyên, Trường Đại học Bách khoa,
Đại học Quốc gia Thành phố Hồ Chí Minh
TÓM TẮT:
Nghiên cứu này được thực hiện nhằm đánh giá hiệu quả quang xúc tác của hạt nano Ag–ZnO trong quá trình phân hủy Rhodamine B (RhB), một chất màu hữu cơ phổ biến trong nước thải. Vật liệu quang xúc tác Ag–ZnO tổng hợp được đặc trưng bằng phương pháp SEM/EDS để khảo sát hình thái bề mặt và thành phần nguyên tố. Kết quả cho thấy hệ quang xúc tác đạt hiệu quả phân hủy cao, với điều kiện tối ưu là lượng Ag–ZnO 0,06 g trong môi trường trung tính (pH 7). Trong điều kiện này, 78% RhB ở nồng độ ban đầu 50 mg/L bị phân hủy sau 120 phút chiếu xạ UV. Phân tích động học cho thấy quá trình phân hủy tuân theo mô hình Langmuir–Hinshelwood giả bậc nhất, với hệ số tương quan cao (R² = 0,9901), chứng tỏ dữ liệu thực nghiệm phù hợp tốt với mô hình đã đề xuất. Hằng số tốc độ biểu kiến được xác định là 0,0123 phút-¹, cho thấy vật liệu Ag–ZnO sau tổng hợp có khả năng phân hủy tương đối tốt so với một số nghiên cứu trước đây. Những kết quả này cho thấy tiềm năng ứng dụng của hạt nano Ag–ZnO như một chất quang xúc tác hiệu quả trong xử lý nước thải chứa thuốc nhuộm.
Từ khóa: Ag-ZnO, thuốc nhuộm, Rhodamine B, tia UV, xử lý nước thải.

